Point-of-care quality wrist and hand imaging: Transforming the way patients experience healthcare
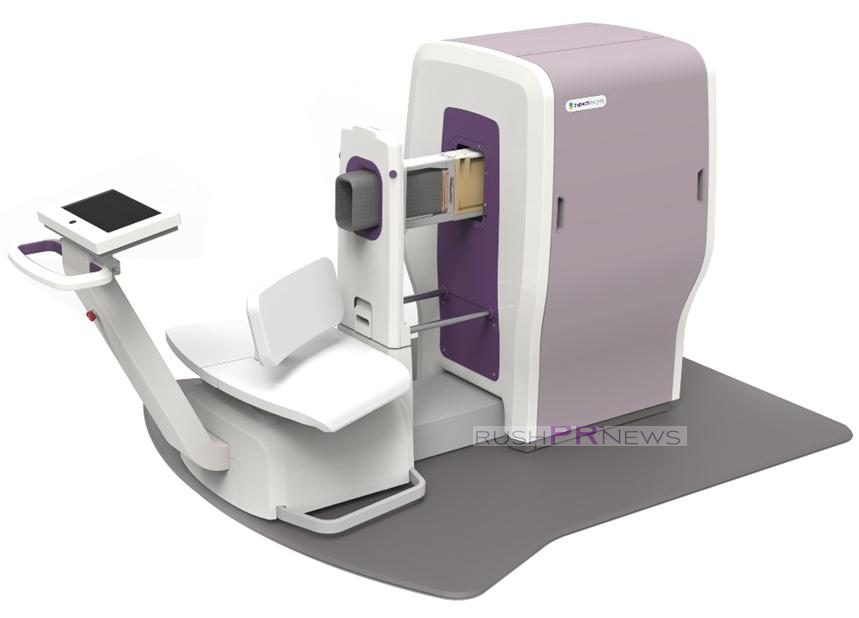
Aspect Imaging, the world leader of compact high-performance complete MRI systems and MR technology, announced that the U.S. Food and Drug Administration (FDA) has cleared the Company’s WristViewTM MRI system for diagnostic imaging of the wrist and hand.
Delivering point-of-care affordable MRI, this latest complete solution from Aspect is a game-changer for the healthcare industry. The WristViewTM makes what was previously only available via a whole body scanner, now fast and effortless straight from doctors’ surgeries and clinics. Patients sit comfortably, inserting only the hand into the magnet. The system operates silently, with an integrated operator station allowing close patient contact throughout the process; ensuring a safer and non-claustrophobic stress-free patient experience.
With the WristViewTM MRI system, high-quality images of the internal structure of the wrist and hand become affordable and easily available from almost any point-of-care. This not only improves medical service efficiency and increases patient satisfaction, but also ensures substantial money savings for clinics and patients alike. Aspect’s non-cryogen technology and passive self-shielded solution only require a single-phase standard electrical socket for installation. A small overall footprint carries minimal installation and running costs with no need for a specially constructed RF-shielded room.
“There is no doubt about the value of MRI-based insights for healthcare and clinical research. Now, with the WristViewTM we’ve overcome the high-cost challenge of more conventional MRI systems in scanning the wrist and hand, opening up a world of potential applications. To give just one example, rheumatology drug trials are increasingly using MRI of the hand area as a secondary endpoint to demonstrate drug efficacy. At Aspect we believe that a dedicated MRI could provide an imaging tool to support managing rheumatoid arthritis symptoms and treatment on a routine basis.” says Uri Rapoport, Aspect Imaging’s Founder and CEO.
Initial WristViewTM MRI system installations are expected during Q3 2016.
About Aspect Imaging:
Aspect Imaging (www.aspectimaging.com) is the world leader in the design and development of complete compact MRI and NMR systems. Our unique technology platform is the backbone for a wide range of products; spanning preclinical, medical, oil & gas and advanced industrial markets. In the pre-clinical research market, the M-series compact one-touch MRI systems enable a wide variety of in vivo applications and research models. The M-series systems are also used to provide 3D MR-based histology, complementing and directing pathology and histology-based analysis. In the medical market, Aspect Imaging has multiple medical programs underway, including highly efficient just-in-place compact MRI systems, such as the FDA-cleared WristViewTM MRI. The company’s novel technology is also used in advanced industrial applications. These include rheology, where the FlowScan™ platform provides real-time, online and quantitative information for process and quality control. In addition, the AI-60 Process Analyzer enables real-time, continuous flow-through stream analysis, with the ability to provide analysis of dense and opaque materials such as crude oil.
For further information, please contact:
Dr. Andrew Lonergan
VP Sales and Marketing, Aspect Imaging